 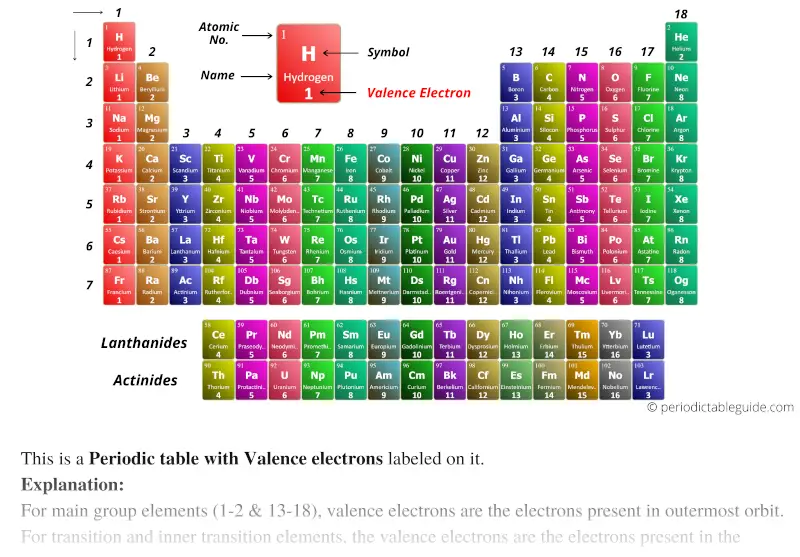
In other words, the number of valence electrons for a transition metal is equal to how many spaces across the periodic table it is. The valence electrons for transition metals are equal to the number of s-electrons plus the number of d-electrons. Which elements do you expect to lose electrons in chemical reactions Which do you expect to gain. For non-transition metals, we count to 8, but for transition metals, we count to 12. Determine the number of valence electrons in each element. Looking at the orbitals explains how valence electrons work for transition metals. /PeriodicTableCharge-BW-56a12db13df78cf772682c34.png)
For example, oxygen has 6 valence electrons, these six electrons fill up the 2s orbital, and partially fill the 2p subshells (2s 22p 4). Ionic charge: When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). Main Group Metals: Charges are determined by the group and. Valence electrons are the electrons in the highest occupied principal energy level of an atom. The shells after (ignoring transition metals) represent the s and p-orbitals. Each element has a charge dictated by whether it is a metal (+ charge) or a nonmetal (- charge). So what does this have to do with our shells? The first "shell" represents the 1s orbital. For example, carbon has an electron configuration of 1s 22s 22p 2. The elements with properties intermediate between those of metals and nonmetals are called metalloids (or. The way we count our electrons is by moving from right to left, starting at the beginning of the table. 2, metals occupy the left three-fourths of the periodic table, while nonmetals (except for hydrogen) are clustered in the upper right-hand corner of the periodic table. F-orbitals start appearing in the lanthanides and actinides (the separated two rows). P-orbitals start appearing in period 2, and d-orbitals start appearing in period 4 (though they start counting at 3). Has 7 subshells, each holding 2 electrons, for a total of 14 electronsīelow is the periodic table with the labeled orbitalsĮach period is its own energy level.Has 5 subshells, each holding 2 electrons, for a total of 10 electrons.Has 3 subshells, each holding 2 electrons, for a total of 6 electrons.Why do the alkali metals generally have a positive charge? 16. Explain why the halogens generally have a negative charge. What is the general trend for increasing metallic character? 15. ion, an atom that has gained or lost an electron, therefore ending up with a charge energy level numbers, found by the number of the periods valence electron. What is the general trend from left to right across a period for electronegativity? How can you explain this trend? Which element has the highest electronegativity? 14. With the exception of groups 312 (the transition metals), the units digit of the group number identifies how many valence electrons are associated with a neutral atom of an element listed under. This creates a positively charged cation due to the loss of electron. This sodium molecule donates the lone electron in its valence orbital in order to achieve octet configuration. In ionic bonds, the net charge of the compound must be zero. In your own words, define electronegativity. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. The charges on the anion and cation correspond to the number of electrons donated or received. Describe the properties attributed to most nonmetals. Describe the properties attributed to most metals. Which seven elements from the periodic table are metalloids? What are the properties of a metalloid? What group numbers contain metalloids? 11. Which group (name and number) from the periodic table contains atoms with a stable outer electron configuration? Why are these elements so stable? 10. So, their ionic charge is usually the same. In what column of the periodic table would you expect to find atoms with 7 valence electrons? List one of these atoms. On the periodic table, elements in the same group have the same number of valence electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
